 
(also due to the screening effect Z eff of 1s with no screening is > than 2s ) are all incorrect. (iii) Z eff for an electron in 1s orbital is the same as Z eff for an electron in a 2s orbital. (in case of oxygen atom this is not possible as energy of 2p orbital is > 2s orbital. A radical (often, but unnecessarily called a free radical) is an atom or group of atoms that have one or more unpaired electrons. In this video well look at the atomic structure and Bohr model for the Oxygen atom (O). (ii)An electron in the 2s orbital has the same energy as an electron in the 2p orbital. (cannot be because 2s and 2p orbital electrons have different screening effect and Z eff of 2s orbital is > than 2p orbital as 2s is closer to the nucleus.) (i) Z eff for an electron in a 2s orbital is the same as Z eff for an electron in a 2p orbital. Hence, the concept effective nuclear charge is introduced which will also be lowered due to the partial screening or shielding of the positive charge on the nucleus by the inner shell electrons.Due to the electrons in the inner shells (nearest to the core), the electron in the outer shell is unable to experience the full positive charge of the nucleus ( Z e).The value of this Z eff depends on the shielding or screening effect of certain orbital electrons.O In the case of multi-electron atoms (O), Z eff is the effective nuclear charge which is the net positive charge of the nucleus, faced by the outer shell (valence) electrons. m s and opposite spins thus, cancelling each other. 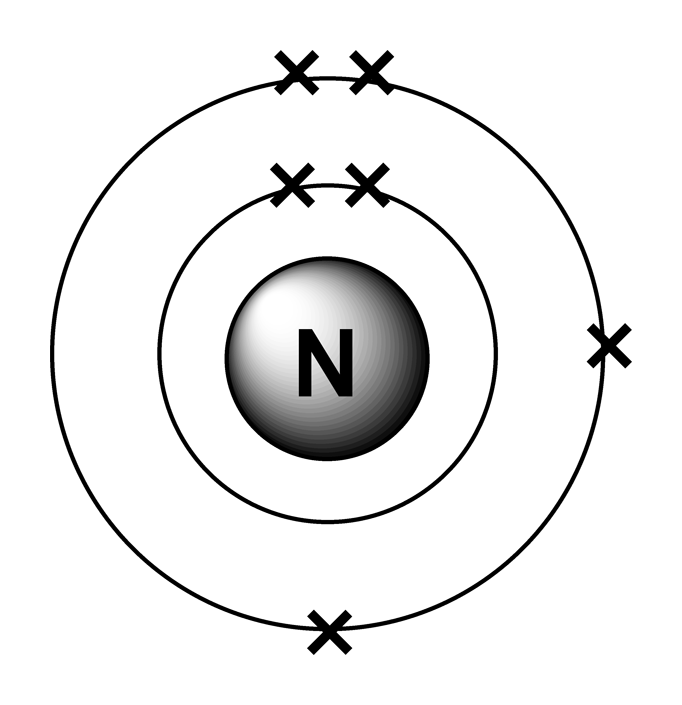
And for an orbital to hold two electrons each of the 2 electrons should be of same quantum numbers i.e. + 1/2and – 1/2which are the 2 spin states ( more than two spin states cannot be held in the orbital). Kerala online shopping for electronics products at best & lowest prices exclusive at Oxygen Buy online from the most trusted and largest digital store in. When the hydrogen and oxygen each share 1 electron with each other, then all the. Oxygen is a highly reactive element that promotes rapid combustion and is often used in industrial applications.O Spin quantum numbers or m s can only take 3 values, i.e. The oxygen has 6 electrons in it s valence shell and the hydrogen each has 1. Oxygen is colorless, odorless, and tasteless in its gaseous form, and condenses to pale blue liquid and solid forms. (a) Molecular orbital diagram of triplet and singlet oxygen (b) distinctive reshaping of the wavefunction for the 3 g (left) states and 1 g (right). Accounting for one-fifth of the earth’s atmosphere, oxygen combines with most elements and is a component of thousands of organic compounds. 
Oxygen is critical for life on Earth, produced by plants during photosynthesis and necessary for aerobic respiration in animals. The Oxygen Evolving Center (OEC) of Photosystem II Watch on Photosynthesis is the process by which plants make energy from the use of chlorophyll and light. 
In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: the change in energy (in kJ/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Oxygen, the "elixir of life", was discovered by Joseph Priestly and Carl Wilhem Scheele independently of each other in the 1770’s. Electron affinity of Oxygen is 141 kJ/mol. Discoverer: Joseph Priestley/Carl Scheele 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
